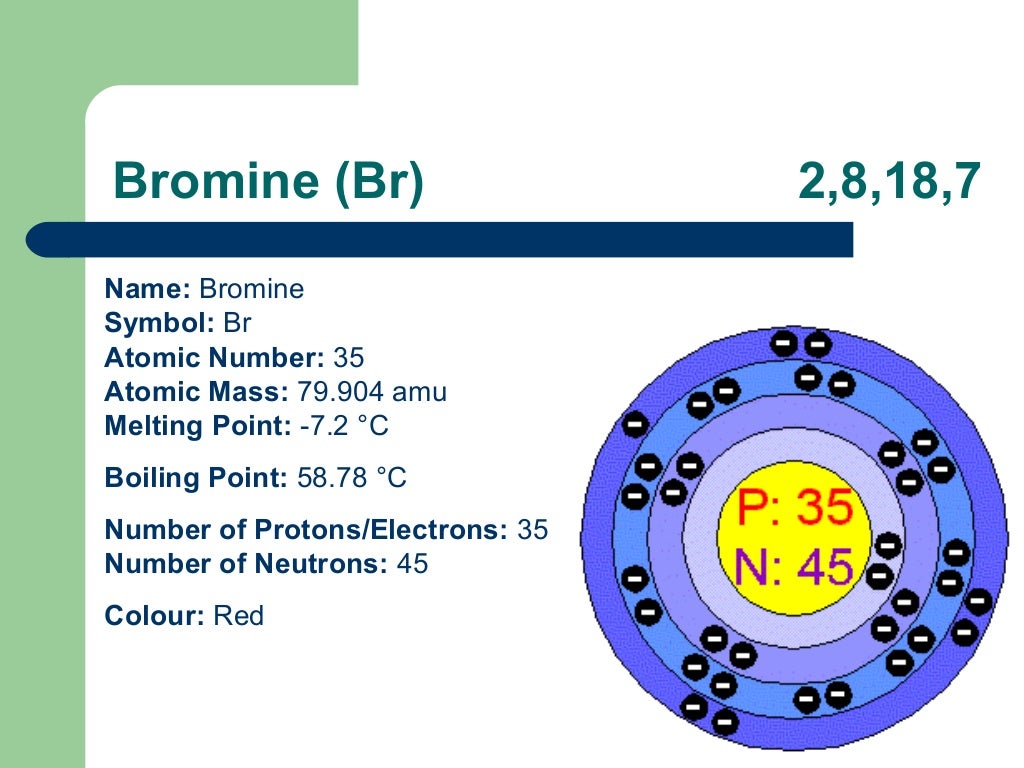
Iodine is the least reactive halogen (besides astatine which is often ignored because it is extremely rare). The halogens get less reactive – fluorine, top of the group, is the most reactive element known.The smallest halogen, fluorine, is the most electronegative element in the periodic table. Electronegativity decreases down the group.The color of the halogens gets darker – fluorine is pale yellow, followed by green chlorine, brown/purple bromine and purple iodine.The melting and boiling point gets higher – starting as gases, bromine is a liquid while iodine is a solid.As you go down the group, the properties of the elements change in the following ways:.They do not conduct heat or electricity.Halogens in elemental form are relatively toxic, reactive substances.They have relatively low melting and boiling points compared to other non-metals (except the noble gases).Halogen ions will usually have a single negative charge (X - ), where they are known as halides. They have a valence of 1 and form covalent bonds with non-metals atoms, or ionic bonds with metal atoms.They are non-metals stable as diatomic molecules (this means at room temperature and pressure, they exist as molecules made of two atoms, e.g.The halogens have the following properties:.As they are reactive elements, they were known and studied in their compounds before being isolated in their reactive, toxic elemental forms. Just like with the alkali metals, the halogens are another example of a well-studied group of elements which display trends in their common properties as you go down the group.The number of outer shell electrons dictates the chemical properties of an element. We have already seen that the Periodic Table is arranged, top-left to bottom-right, by proton number and number of outer shell electrons.To know the key reactions of the halogens.To apply knowledge of electronic structure and bonding to explain the trends in halogens.To understand the trend in properties found in the halogens.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
